10 Apr Selsaf®, a natural source of selenium with dual protection and dual benefits

In 1817, selenium (Se) was isolated for the first time by Berzelius. At that time little was known about its biological function, until its nutritional requirement was revealed about 140 years later by Schwarz and Foltz in 1957 (Mehdi et al., 2013). Since then, Se has been recognized as an essential trace element and its supplementation to farm animals has become a common practice worldwide, as Se deficiency increases susceptibility to various diseases and decreases productive and reproductive performances of farm animals (Lyons et al., 2007).
The essence of Se to exert its positive effects lies in the construction of the 21th amino acid selenocysteine (SeCys). SeCys can be found at the active site of a wide range of proteins, also called selenoproteins, which play important roles in the antioxidant defense and immune function. One of the most importent selenoproteins identified is glutathione peroxidase (GPx), which functions as an antioxidant by directly reducing hydrogen peroxides (H2O2) and phospholipase A2 cleaved lipid hydroperoxides and thus reducing the level of lipid and protein oxidation in the cell (Brown and Arthur, 2001).
Selenium metabolism in yeast and animals
Nowadays, farm animals can be supplemented with Se under an inorganic or an organic form. These inorganic forms are mainly mineral salts such as sodium selenite (SS) or selenate, while organic forms can be found as synthetic selenomethionine (SeMet) or as selenized yeasts rich in natural Se components. These selenized yeasts can be produced in different ways, but the highly consistent selenized yeast ‘Selsaf®’ is obtained due to the specific cultivation of a proprietary Saccharomyces cerevisiae (CNCM I-3399) strain on a medium enriched with SS. During the growth of the yeast, SS will be transformed to hydrogen selenide (H2Se). H2Se is an intermediate metabolite used by the yeast to synthetize different organic selenomolecules such as the seleno amino acids selenomethionine (SeMet) and selenocysteine (SeCys), but also many other seleno-active components (Kieliszek et al. 2015). Consequently, due to the standardized production procedure, Selsaf® contains 2 natural organic Se fractions which are easily absorbed in the animals’ intestine I) 63% SeMet and II) 36% SeCys and other active selenocomponents. In the first fraction, SeMet closely resemblances the amino acid methionine (Met), as the only difference between the 2 is the substitution of the sulfur atom by a selenium atom in SeMet. This will lead to the non-specific incorporation of SeMet in the animal’s body proteins. As such, a pool of Se under the form of SeMet is created in animal proteins found in eggs and milk, leading an efficient transfer of Se to offspring. Additionally, SeMet is also incorporated and stored in the muscles where it can be addressed in stressful times. Under stress conditions, SeMet will be released from this reserve pool in the muscle due to catabolism and liberated SeMet can be converted to H2Se for the construction of animal SeCys in the seleno-enzymes (Schrauzer, 2000). To reduce this muscle catabolism under stress conditions and thus the influence of stress on zootechnical performance of farm animals, Selsaf® harbors a directly available source of active seleno-components which can be used immediately for the conversion to H2Se and subsequently for the formation of seleno-enzymes like GPx.
Interest of Selsaf® for health
The capacity Selsaf® to increase the activity of seleno-enzymes, like GPx, and thus its ability to improve the anti-oxidative status of varies animal species, compared to non Se supplemented animals (NC) was previously demonstrated and is represented for dairy cows in figure 1 (unpublished data).
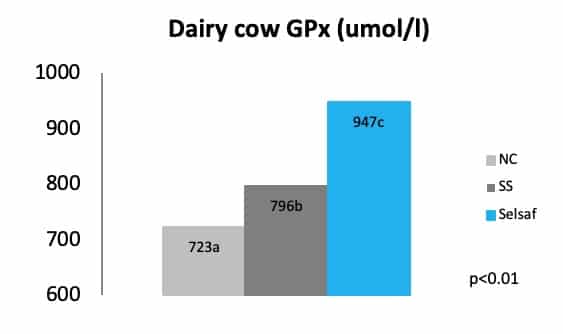
Additionally, a trial in beef cattle recently demonstrated that supplementation of Selsaf® to these animals is able to increase the antiradical capacity of the whole blood when samples are exposed to a free radical attack. This antiradical capacity is in fact the measurement of the time necessary for the free radicals to hemolyze 50% of the erythrocytes. The data in figure 2 clearly show that free radicals need a significantly longer time to destroy erythrocytes in samples from animals receiving Selsaf® compared to SS (unpublished data). This means that Selsaf® is more efficient in improving the antioxidant capacity compared to inorganic sources of selenium (unpublished data).
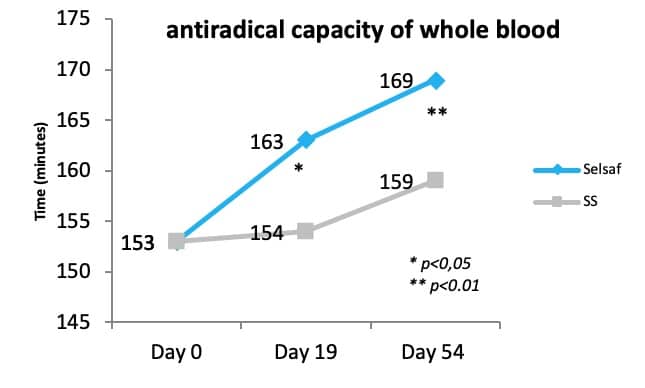
As oxidative stress is very often related to immunity Selsaf® raises not only the antioxidant capacities of animals but influences their immune system. As such, Selsaf® supplemented animals developed a higher number of neutralizing antibodies in serum after vaccination with a bovine herpesvirus 1 (BHV1) vaccine administrated on day 0 and 19 of the experiment (Fig. 3) (unpublished data).
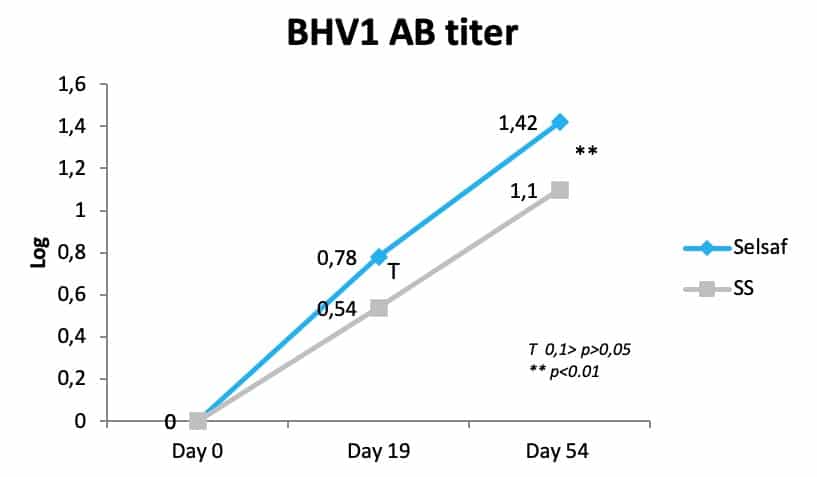
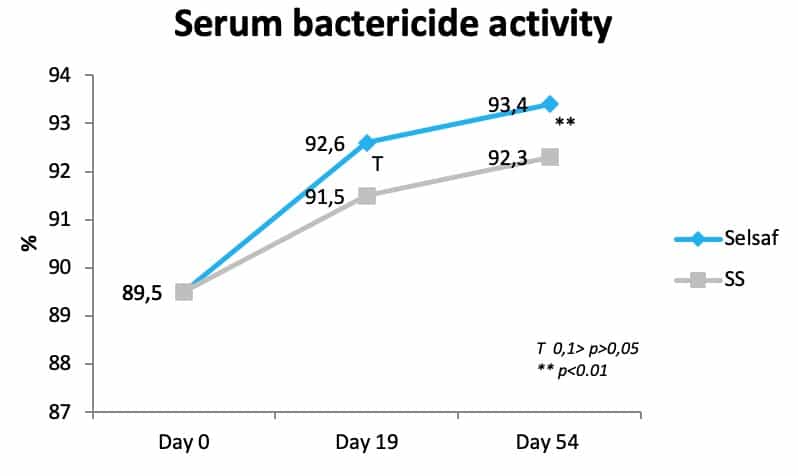
Next to increasing the specific immunity after vaccination, Selsaf® was also capable of improving the innate immunity as evidenced by measuring the bactericide activity of serum, which is the capacity of serum to inhibit the activity of some bacterial strains and thus characterizes the non-specific immune system. In bovine species, a capacity below 90% indicates an increased sensitivity to pathogenic infections. Consequently, data show that at the beginning of the trial, so immediately after transportation and before Se supplementation had started, all animals were highly sensitive to infection (Fig. 4) (unpublished data). After starting with Se administration, animals in the Selsaf® group exhibited a stronger and faster recovery of their serum bactericide activity and where thus better able to resist pathogens.
Practical consequences for animal performances
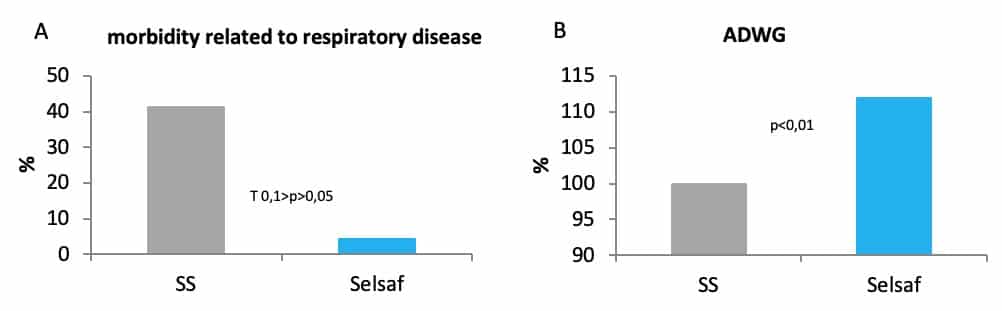
As a matter of fact, in cattle respiratory pathologies are often of bacterial origin and generally appear in a range period between 6 to 10 days after exposure to a stressing event like transport (Hjerpe, 1983). The fast and strong recovery of the non-specific immune system after transportation with Selsaf® supplementation, was reflected in the incidence of morbidity related to respiratory disease which decreased from 41,2% in control group supplemented with SS to only 4,3% in animals receiving Selsaf® (Fig. 5A). Furthermore, the ability of Selsaf® supplemented animals to cope with stress was also twinned in the growth performance as cattle from the control group with SS did not recover quickly their growth deficit accumulated during the period of convalescence (Fig. 5B) (unpublished data).
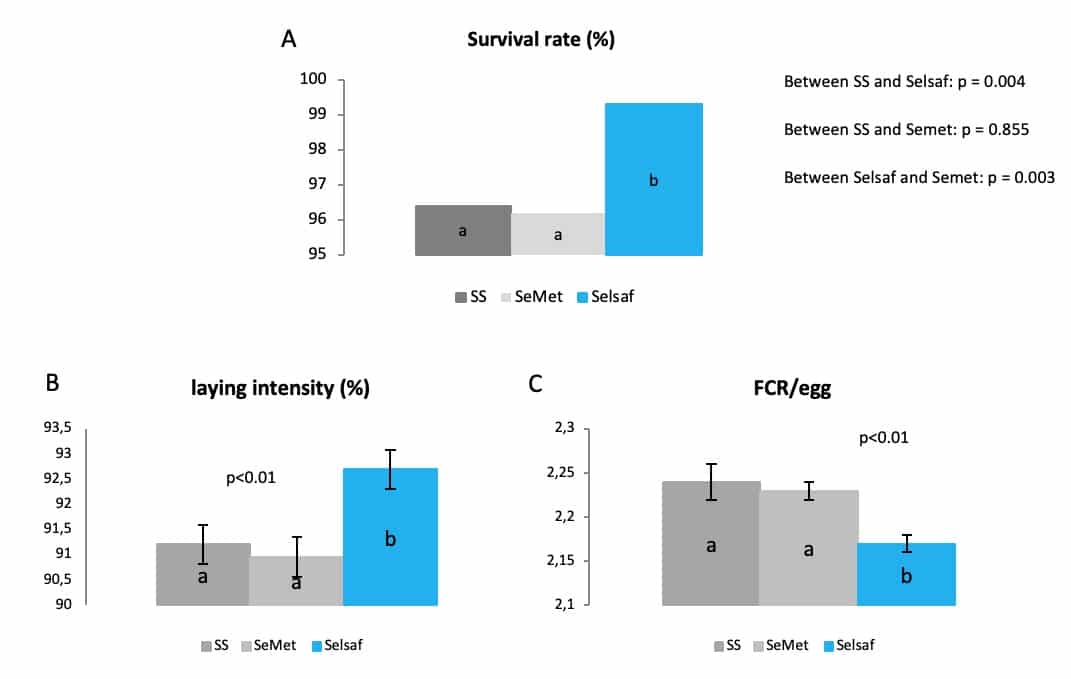
The influence of Selsaf® on animals’ general health and performance was also demonstrated in poultry. In a first trial, laying hens were supplemented for 32 weeks (from 18 to 50 weeks of age) with different Se sources, going from SS, Selsaf® to a source of synthetic SeMet. As shown in figure 6 (A-C). The results clearly show that Selsaf® was able to reduce the mortality (A) of the layers while improving their laying intensity (B) and feed conversion ratio (FCR) per egg (C), compared to the other Se sources.
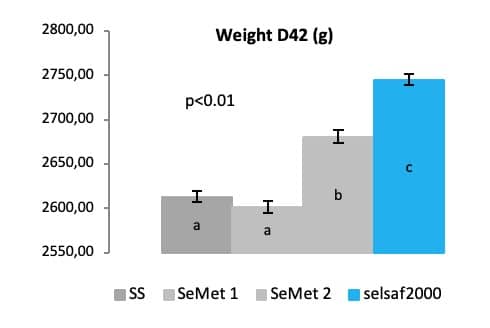
In a second poultry trial done in broilers, additional evidence was gathered to evince that Selsaf® is more capable at improving broiler performance compared to other Se sources (inorganic and synthetic SeMet) as demonstrated by their final body weight at 42 days of age (Fig. 7) (Unpublished data).
Consequently, it appears that Selsaf® is not only capable of ameliorating the antioxidant and immune function of animals but that it also comprises benefits for farmers by reducing morbidity/mortality of the animals and by improving their performance.
Practical consequences for consumers
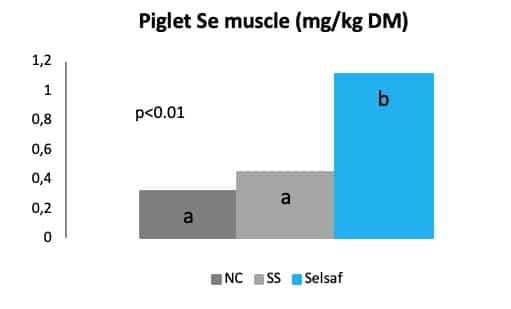
Finally, not only farmers seem to profit from the supplementation of their animals with Selsaf® but also consumers might experience some positive effect when eating end-products derived from animals supplemented with Selsaf®. As such, due to the presence of SeMet, Se is deposited in several animal derived products e.g. meat, milk, egg (Fig. 8) which are frequently consumed by humans. In this way, the consumption of animal products might lead to acquisition of natural Se in humans and consequently an improved oxidative status with better resistance to pathogenic infections.
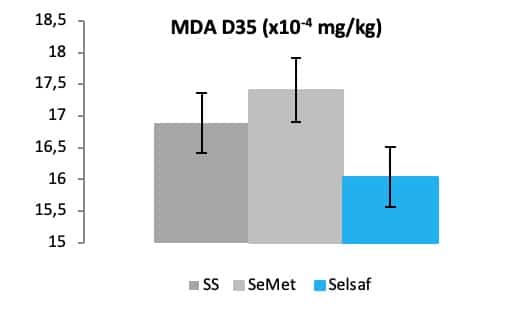
Additionally, it is well known that eggs are susceptible to lipid peroxidation due to the presence of polyunsaturated fatty acids (Mohiti-Asli et al., 2008). The oxidation level lipids can be demonstrated by the measurement of malondialdehyde (MDA), a secondary oxidation product formed during lipid oxidation. As such, the concentration of MDA can function as a parameter to assess the influence a certain Se source on lipid oxidation. As a matter of fact, foods appropriate for consumption should present lipid oxidation values below 3 mg MDA/kg of sample with an upper limit of 7-8 mg MDA/kg (Faitarone et al., 2016).
Consequently, by using this parameter, it was recently demonstrated that Selsaf® is able to reduce the lipid oxidation in eggs during 35 days of storage (Fig.9), meaning that their shelf life would increase by supplementing hens with Selsaf®.
In meat the oxidation of lipids and proteins is the most important parameter for the appearance of meat rancidity. On one hand, the oxidation of proteins in meat might decrease the activity of proteases and increases the myofibrillar protein cross linking finally resulting in tougher meat texture while lipid oxidation on the other hand might interferes with the integrity of the cell membrane leading to a lower water holding capacity and the formation of volatile oxidation products interfering with odor and flavor (Rossi et al., 2015).
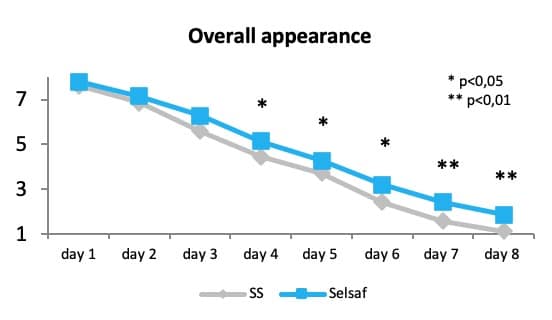
Recent data indicate that supplementation of Selsaf® to beef cattle might affect the color, odor, surface wetness and overall appearance of the meat during storage. Data clearly show that the color score was higher in the Selsaf® supplemented group from the 4th day and that the other score was higher on the last 2 days while surface wetness was higher starting from the 5th day of storage. Consequently, the overall appearance of the meat was increased from the 4th day of storage by the supplementation of Selsaf® to the animal (Rossi et al., 2015) (Fig. 10).
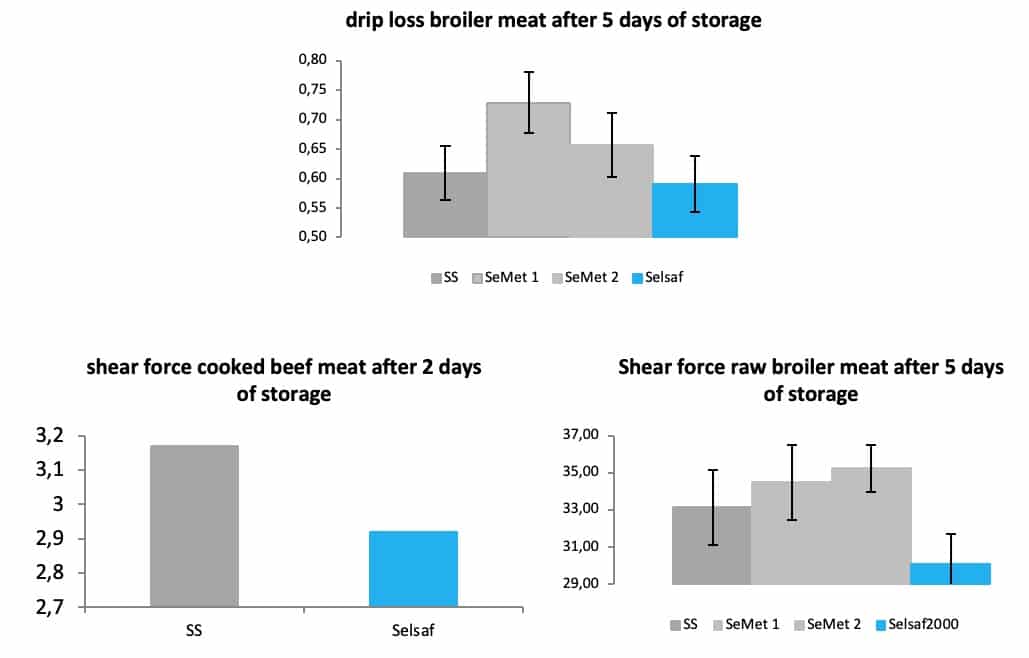
This trial (Rossi et al 2015) and a recent trial performed in broilers (unpublished data) also showed that not only visual aspects of meat were affected by the supplementation of animals with Selsaf® but that also the water holding capacity (Fig. 11A) and the tenderness of the meat (Fig; 11B-C) were positively influenced to a higher degree compared to other Se sources even synthetic SeMet products. Water holding capacity and meat tenderness are respectively measured by means of drip loss and shear force i.e. the strength or force necessary to rupture a meat sample, indicating that the lower the force needed to rupture the meat the more tender the meat will be.
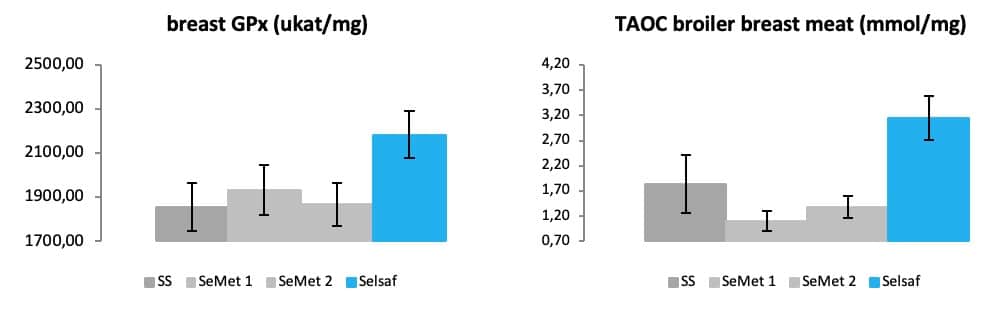
These results are most a likely a consequence of the higher antioxidative status reached in meat when animals were supplemented with Selsaf® compared to the other Se sources as indicated by the measurements of GPx and total antioxidant capacity (TAOC) (Fig. 12A-B) (unpublished data). In fact, TAOC is the measurement of the combined non-enzymatic antioxidant capacity which thus provides information concerning the overall capability to counteract reactive oxygen species (ROS) and resist oxidative damages.
Conclusion
In conclusion, Selsaf® is a natural source of selenomethionine and selenocysteine offering a dual protection against both oxidative and pathogenic stress for the animal but also exhibiting benefits for farmers and consumers by sustaining animal health and enhancing zootechnical performance for farmers and raising consumers’ satisfaction while preserving food quality during shelf life.
References
Brown K.m. and Arthur J. R. (2001). Selenium, selenoproteins and human health: a review. Public Health Nutr. 4(2B):593-599.
Faitarone A.B.G., Garcia E.A., Roça R.O., Andrade E.N., Vercese F. and Pelicia K. (2016). Yolk color and lipid oxidation of the eggs of commercial white layers fed diets supplemented with vegetable oils. Bras. Cienc. Avic. 18 doi.org/10.1590/1516-635X1801009-016.
Kieliszek M., Blazejak S. and Gientka I. (2015). Accumulation and metabolism of selenium by yeast cells. Appl. Microbiol. Biotechnol. 99: 5373-5382.
Hjerpe C.A. (1983). Clinical management of respiratory disease in feedlot cattle. Vat. Clin. North. Am. Large. Anim. Pract. 5(1): 119-142.
Lyons M.P., Papazyan T.T. and Surai P.F. (2007). Selenium in food chain and animal nutrition: Lessons from nature. Asian-Aust. J. Anim. Sci. 20(7): 1135-1155.
Mohiti-Asli M., Shariatmadari F., Lotfollahian H. and Mazuji M.T. (2007). Effects of supplementing layer hen diets with selenium and vitamin E on egg quality, lipid oxidation and fatty acid composition during storage. Can. J. Anim. Sci. 475-483.
Mehdi Y., Hornick J.L., Istasse L. and Dufrasne I. (2013). Selenium in the environment, metabolism and involvement in body functions. Molecules 18: 3292-3311.
Rossi C.A.S., Compiani R., Baldi G., Bernardi C.E.M., Muraro M., Marden J.-P. and Dell’Orto V. (2015). The effects of different selenium sources during the finishing phase on beef quality. Journal of Animal and Feed Sciences, 24, 2015, 93–99.
Schrauzer G.N. (2000). Selenomethionine: a review of its nutritional significance, metabolism and toxicity. J. Nutr. 130: 1653-1656.







Sorry, the comment form is closed at this time.